Table of Contents
Aicardi-Goutières syndrome
AGS is distinct from the similarly named Aicardi syndrome (characterized by absence of a brain structure (corpus callosum), and spinal, skeletal, and eye abnormalities
Aicardi-Goutieres syndrome (AGS) is a genetic encephalopathy characterized by the inappropriate induction of a type I interferon-mediated immune response and usually results in severe cognitive and physical morbidities. It is named after Jean Aicardi and Francois Goutières who first described the condition in 1984[1] in patients who had presented with early onset encephalopathy, basal ganglia calcification, and persistent lymphocytosis in the cerebrospinal fluid. The condition has subsequently been shown to be both genotypically and clinically heterogenous.
Clinical features
There are two distinct forms of the syndrome:
- Early onset
- severe
- affects about 20 percent of all infants who have AGS
- symptoms present at birth with hepatomegaly, splenomegaly, elevated liver enzymes
- irritability & poor feeding (mimicking congenital viral infection)
- late-onset form
- less impact upon neurological function
- symptoms after the first weeks or months of normal development
- progressive decline in head growth
- muscle weakness & spasticity
- moderate to severe cognitive and developmental delay
- Symptoms last for several months, and include irritability, inconsolable crying, intermittent fever, seizures, and loss of developmental skills
- Skin manifestations include puffy swelling on the fingers, toes, and ears that resemble chilblains, acrocyanosis, periungual erythema, or necrotic lesions of the toes, fingers, and outer helix
- startle reaction to sudden noise
- no further worsening of the disease once the symptoms lessen
- 50% - 75% of patients with AGS develop focal and generalized seizures, which are sometimes refractory to treatment[2].
However the clinical features can be quite heterogenous. The neurological dysfunction is not always severe and need not even be present at all. Other features originally described including microcephaly, intracranial calcification, white matter changes and CSF lymphocytosis are not always present[3].
Genetics
AGS is a genetically heterogeneous disease. Most cases of AGS are inherited in an autosomal recessive manner. However it can also be autosomal dominant with heterozygous mutations in TREX, ADAR, or IFIH1 genes. In some cases AGS-associated mutations show incomplete penetrance with children in the same family with the same mutations showing markedly different neurological and developmental outcomes[4]. To date AGS has been associated with mutations of seven different genes.
| Type | OMIM | Gene | Locus | Frequency | Inheritance | Phenotypes | Protein Function |
|---|---|---|---|---|---|---|---|
| AGS1 | 225750 | TREX1 | 3p21.31 | 23% (1% dominant) | AR (rare AD cases reported) | AGS, FCL, RVCL, SLE | 3′-5′ exonuclease with preference for ssDNA |
| AGS2 | 610181 | RNASEH2B | 13q14.3 | 36% | AR | AGS | Non-catalytic component of RNase H2 complex |
| AGS3 | 610329 | RNASEH2C | 11q13.1 | 12% | AR | AGS | Non-catalytic component of RNase H2 complex |
| AGS4 | 610333 | RNASEH2A | 19p13.2 | 5% | AR | AGS | Catalytic component of RNase H2 complex Acts on RNA portion of RNA/DNA hybrids and removes ribonuceotides embedded in DNA |
| AGS5 | 612952 | SAMHD1 | 20q11.23 | 13% | AR | AGS, FCL | dNTP triphosphohydrolase triphosphatase |
| AGS6 | 615010 | ADAR | 1q21.3 | 7% (1% dominant) | AR, AD | AGS, DSH | Hydrolytic deamination of adenosine to inosine in dsRNA |
| AGS7 | 615846 | IFIH1 | 2q24 | 3% (all dominant) | AD | AGS |
The late onset form is observed most commonly in the context of mutations in RNASEH2B, SAMHD1 and ADAR1 while mutations in TREX1, RNASEH2A and RNASEH2C, mimic the sequelae of congenital, transplacentally acquired infection[3].
Pathophysiology
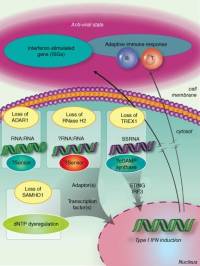
Both AGS and many placentally acquired viral infections are characterized by the production of high levels of IFNα. Astrocyte-specific chronic overproduction of IFNα in transgenic mice has been shown to recapitulate the neuropathologic findings seen in AGS[5]. AGS shows a phenotypic overlap with some aspects of systemic lupus erythematosus (SLE) where an equivalent type I interferon-mediated innate immune response is triggered by self nucleic acids. Current experimental evidence suggests that the nucleases defective in AGS are involved in removing endogenously produced nucleic acid species, and that a failure of this removal results in activation of the immune system[6].
The actual pathomechanisms that lead to CNS damage in AGS patients are unknown. In a mouse model transgenic for a glial fibrillary acidic protein-interferon-alpha fusion protein (GFAP-IFN-<m>alpha</m> mice) where IFN <m>alpha</m> is expressed in an astrocyte-restricted fashion, transgenic animals develop a progressive inflammatory encephalopathy with neuropathologic features similar to AGS suggesting a neurotoxic role of IFN<m>alpha</m> in the developing brain[5].
Interferonopathies comprise an expanding group of monogenic diseases characterised by disturbance of the homeostatic control of interferon (IFN)-mediated immune responses[7].
Diagnosis
- AGS is difficult to diagnose since many of the symptoms are similar to those of other disorders
- Diagnosis is based on the clinical symptoms of the disease
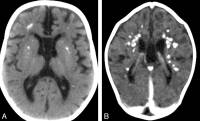
- Radiological abnormalities include basal ganglia calcification (usually bilateral) involving globus pallidus, putamen, caudate nucleus, and dentate nuclei. It may also extend to the periventricular white matter; increased signal on T2 weighted brain MRI particularly in the frontotemporal region along with cerebral atrophy[8][9].
- Cerebrospinal fluid (CSF) shows increased levels of lymphocytes indicating chronic lymphocytosis in the absence of evidence of infection.
- Raised interferon <m>gamma</m> in CSF
Treatment
- Treatment in the early stages of the disease might result in attenuation of the associated inflammation and consequent tissue damage
- Treatment may be discontinued once the subacute encephalopathic period subsides
- Treatment may have to be continued beyond the subacute encephalopathic phase in cases where chilblains are a particular problem
- immune-modulating therapies have been tried empirically including prednisone with azathioprine, intravenous (i.v.) methylprednisolone with i.v. immunoglobulin (IVIG), methylprednisolone and IVIG alone with variable results.
- Anti-interferon alpha antibody therapy: blocking interferon alpha activity using monoclonal antibodies is currently under consideration and clinical trials are currently underway in SLE[10].
- Reverse transcriptase inhibitors (RTIs) (compounds that can potentially disrupt the replication cycle of both exogenous retroviruses and endogenous retro-elements) are of potential interest in treating AGS. RTIs are already prescribed in children and adults with HIV-1 infection[3].
- Agents that deplete B cells are also potential therapeutic agents since AGS is associated with lymphocytes and autoantibody production
- Other agents of interest include drugs that target autoreactive T cells such as Mycophenolate Mofetil
References
[PMID: 6712192] [DOI: 10.1002/ana.410150109]
[PMID: 24011626] [DOI: 10.1016/j.ejpn.2013.07.005]
[PMID: 23607857] [PMCID: 3898548] [DOI: 10.1111/cei.12115]
[PMID: 25604658] [PMCID: 4382202] [DOI: 10.1002/ajmg.a.36887]
[PMID: 9794439]
[PMID: 19808788] [PMCID: 2758706] [DOI: 10.1093/hmg/ddp293]
[PMID: 29773266] [DOI: 10.1016/j.berh.2017.12.002]
[PMID: 26581299] [PMCID: 4731289] [DOI: 10.1212/WNL.0000000000002228]
[PMID: 19628626] [PMCID: 7051307] [DOI: 10.3174/ajnr.A1694]
[PMID: 20948567] [PMCID: 2950308] [DOI: 10.4061/2009/374312]