Aicardi syndrome
A rare neurodevelopmental disorder characterized by the classic triad of agenesis of the corpus callosum (total or partial), central chorioretinal lacunae and infantile spasms that affects almost exclusively females & rarely in 47 XXY males. However Aicardi syndrome is now recognized as a more complex, pleiotropic disorder with an expanded spectrum of phenotypical features.
Clinical features
- Agenesis of the corpus callosum
- Infantile spasms
- Chorioretinal lacunae
- Microcephaly, heterotopias
- polymicrogyria
- Cerebellar dysgenesis
- Microphthalmia , coloboma
- Costovertebral defects
- Facial asymmetry
- Intellectual disability
- developmental delay
- hypotonia
- Focal seizures
Seizures
Infantile spasms are the most characteristic seizure type in Aicardi syndrome[1]. Most seizures appear early, at 3–4 months of age, and are often asymmetric or even unilateral. With increasing age, the seizure pattern evolves and focal seizures become more frequent[2]. EEG features include asynchronous multifocal epileptiform abnormalities with burst suppression and dissociation between the two hemispheres[3].
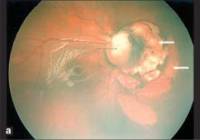
Chorioretinal lacunae
Central Chorioretinal lacunae (CRL) represent a defect of the pigment epithelium and choroid. CRL are pathognomonic for Aicardi syndrome and are seen in all patients (see figure[4]).They are well-defined, multifocal pale areas with minimally pigmented borders, and are usually found in the peripapillary area of the posterior pole. They do not change or evolve over time. CRL do not affect vision unless the central foveal area is involved.
Optic nerve anomalies are present in the majority of affected individuals, with coloboma being the most common. Other abnormalities include asymmetric microphthalmia, morning glory discs, abnormal retinal vessels and cysts and often result in significant visual impairment.
Dysmorphology
Aicardi syndrome is associated with distinctive facial features including prominent premaxilla, upturned nasal tip, decreased angle of the nasal bridge, and sparse lateral eyebrows.
Other abnormalities reported include hemivertebrae, block vertebrae, fused vertebrae, and missing, malformed or fused ribs[5] hand abnormalities including camptodactyly, proximal placement of the thumb, and hypoplasia of the fifth finger[6][7].
Diagnostic criteria
Sutton et al. (2005) proposed the following modified diagnostic criteria[7]:
- The presence of all three classical features (classic triad) is diagnostic for Aicardi syndrome
- The presence of two classical features in addition to at least two major or supporting features is strongly suggestive of a diagnosis of Aicardi syndrome
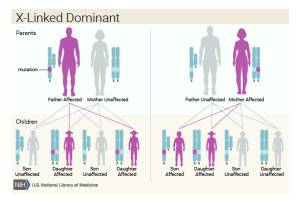
Genetics
- Nearly all reported cases of Aicardi syndrome are sporadic and in females and believed to result from new gene mutations
Neuroimaging
Abnormalities identified on MRI are characteristically[9]
- polymicrogyria that was predominantly frontal and perisylvian and often associated with underopercularization
- Periventricular nodular heterotopias
- single or multiple intracranial cysts
- Cerebellar abnormalities
- tectal enlargement
Differential Diagnosis
Treatment
- seizure management. Seizures are often refractory
- physical, occupational, speech therapy
- vision
- management of vertebral abnormalities including scoliosis
Prognosis
- majority of children are unable to sit independently, walk or talk
- The 5-, 10- and 20-year survival rates are 90%, 80% and 50%, respectively[10]
History
In 1965 French pediatric neurologist and epileptologist Jean Aicardi described 8 children with infantile spasm-in-flexion, total or partial agenesis of the corpus callosum, and variable ocular abnormalities[11]. Further 7 patients were described in 1969[12] and the name “Aicardi syndrome” was established in 1972, by Dennis and Bower. [13].
References
[PMID: 15737696] [DOI: 10.1016/j.braindev.2003.11.011]
[PMID: 17621479] [DOI: 10.1177/0883073807300298]
[PMID: 16967367] [DOI: 10.1055/s-2006-924486]
[PMID: 19384023] [PMCID: 2683450] [DOI: 10.4103/0301-4738.49403]
[PMID: 4314028]
[PMID: 2773986] [DOI: 10.1002/ajmg.1320320405]
[PMID: 16158440] [DOI: 10.1002/ajmg.a.30963]
[PMID: 26091538] [DOI: 10.1167/iovs.14-16261]
[PMID: 18925666] [PMCID: 2597151] [DOI: 10.1002/ajmg.a.32537]
[PMID: 4314028]
[PMID: 4624502] [DOI: 10.1111/j.1469-8749.1972.tb02604.x]

Discussion