Ohtahara syndrome
Ohtahara syndrome is a rare electroclinical epilepsy syndrome with onset in the neonatal period. It is also known as early infantile epileptic encephalopathy (EIEE) or early infantile epileptic encephalopathy with burst suppression pattern. It is named after Shunsuke Ohtahara, the Japanese child neurologist who first described the syndrome in 1976 [1].
Epidemiology
- Incidence has been estimated at 1/100 000 births in Japan and 1/50,000 births in the U.K[2]
Pathophysiology
Majority of Ohtahara syndrome patients have an underlying structural abnormality. The associated structural abnormalities include hemimegalencephaly, porencephaly, dentato-olivary dysplasia[3], agenesis of the corpus callosum, agenesis of the mamillary bodies[4], cortical dysplasia and neuronal migration disorders. Ohtahara syndrome has also been reported to be caused by underlying metabolic conditions including nonketotic hyperglycinemia, cytochrome C oxidase deficiency, pyridoxine dependency, carnitine palmitoyltransferase deficiency, Leigh encephalopathy, biotinidase deficiency and mitochondrial respiratory chain complex I deficiency.
Genetics
Although structural abnormalities are the most common underlying finding in Ohtahara syndrome, genetic mutations have also been increasingly reported. Mutations known to be associated with Ohtahara syndrome include syntaxin binding protein 1 (STXBP1) gene[5], Aristaless-related homeobox (ARX) gene, solute carrier family 25 (SLC25A22) gene. However these genetic abnormalities are also related to brain structural abnormalities. For e.g ARX gene mutations can result in hypoplastic corpus callosum, cerebral atrophy, abnormal cavum septum pellucidum and abnormal hippocampi and basal ganglia. STXBPI mutations have been associated with brain stem structural abnormalities. The tonic seizures seen in Ohtahara syndrome are thought to be originating in the brain stem[6].
Other genes associated with Ohtahara syndrome include CDKL5; SPTAN1; KCNQ2; ARHGEF9; PCDH19; PNKP; SCN2A; PLCB1; SCN8A; ST3GAL3; TBC1D24; BRAT1
Clinical features
Ohtahara syndrome presents in early infancy, within the first 3 months of age, and often within the first 2 weeks of life[7]. Infants develop tonic spasms that are either generalized or lateralized. The spasms occur either singly or in clusters and are unrelated to the sleep cycle. The spasms can occur several times per day and typically last upto 10 seconds. Some children also develop other seizure types including focal motor seizures, hemiconvulsions, or generalized tonic-clonic seizures.
Electrographic features
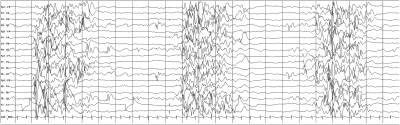
In Ohtahara syndrome electroencephalograms charactaristically show a suppression burst pattern, with bursts of high-amplitude spikes and polyspikes that alternate at a regular rate with periods of electric suppression (see Figure 1). The bursts coincide with the tonic spasms[8]. The pattern typically remains the same during both wakefulness and sleep.
Inter-ictal EEG
- Pseudorhythmic repetitive suppression-burst pattern without physiological rhythms
- Age-related evolution to hypsarrhythmia of West syndrome and then to slow spike-wave patterns of Lennox-Gastaut syndrome.
Ictal EEG
- tonic spasms of variable duration concomitant with the burst phase
- When tonic spasms cluster in intervals of 5 to 10 sec there could be diffuse desynchronization with disappearance of suppression-burst activity or the suppression-burst pattern could become more frequent, diffuse, and of higher amplitude compared to the inter-ictal pattern.
Management
Diagnosis is based on the clinical picture and the electrographic characteristics. Neuroimaging is warranted to look for structural abnormalities.
- EEG
- MRI
- Genetics- CGH Microarray followed by epilepsy gene panel and whole exome sequencing if necessary
Treatment depends on management of the epilepsy.
- Anti-seizure medications
- Vagal Nerve Stimulation (VNS)
- Ketogenic diet
- Epilepsy surgery
Prognosis
- Prognosis is universally poor
- Ohtahara syndrome can evolve to West syndrome and then to Lennox-Gastaut syndrome, or can transition to severe focal epilepsy
- High incidence of mortality & morbidity with permanent cognitive and neurologic deficits
Differential Diagnosis
There is considerable clinical overlap between the two conditions and some authors have suggested that they are part of a spectrum since they have similar pathophysiologic pathways[6].
Support
Aarons Ohtahara E-mail: https://sites.google.com/a/ohtahara.org/ohtahara2/contact-us Website: http://www.ohtahara.org/
Epilepsy Foundation 8301 Professional Place East Suite 230 Landover, MD 20785 Toll-free: 800-332-1000 (24/7 Helpline) Telephone: +1-301-459-3700 Fax: +1-301-577-2684 E-mail: contactus@efa.org Website: https://www.epilepsy.com/ en Español 1-866-748-8008
References
|
2.
a
RESERVED IU–AR. Orphanet: Early infantile epileptic encephalopathy Ohtahara syndrome [Internet]. [cited 2020 Jan 28]. Available from: http://icnapedia.org/s/ohtahara-syndrome
3.
a
Schlumberger, E.et al. Early-infantile epileptic syndrome(s) with suppression-burst: Nosological considerations. in: J. Roger, M. Bureau, C.H. Dravet, F.E. Dreifuss, A. Perret, P. Wolf (Eds.) Epileptic syndromes in infancy, childhood, and adolescence. John Libbey & Company Ltd., London; 1992: 35–42
4.
a
. A case of Ohtahara syndrome with olivary-dentate dysplasia and agenesis of mamillary bodies. Epilepsia. 2001 Jul;42(7):950-3. doi: 10.1046/j.1528-1157.2001.042007950.x.
[PMID: 11488899] [DOI: 10.1046/j.1528-1157.2001.042007950.x] |
5.
a
. De novo mutations in the gene encoding STXBP1 (MUNC18-1) cause early infantile epileptic encephalopathy. Nat Genet. 2008 Jun;40(6):782-8. doi: 10.1038/ng.150. Epub 2008 May 11.
[PMID: 18469812] [DOI: 10.1038/ng.150]
6.
a,
b,
c
. Early-onset epileptic encephalopathies: Ohtahara syndrome and early myoclonic encephalopathy. Pediatr Neurol. 2012 Nov;47(5):317-23. doi: 10.1016/j.pediatrneurol.2012.06.002.
[PMID: 23044011] [DOI: 10.1016/j.pediatrneurol.2012.06.002]
8.
a
. Video/EEG aspects of early-infantile epileptic encephalopathy with suppression-bursts (Ohtahara syndrome). Brain Dev. 2001 Nov;23(7):708-14. doi: 10.1016/s0387-7604(01)00280-7.
[PMID: 11701283] [DOI: 10.1016/s0387-7604(01)00280-7] |


Discussion